Titanium dioxide decreases air pollution
Given that nitrogen oxides contribute decisively to the formation of ozone in the lower layers of the atmosphere (troposphere), the reduction of the concentration of oxides of nitrogen in the air allows expect a decrease of the problems caused by 'smog' in summer. While already at different levels have been adopted concrete measures to reduce emissions of harmful substances, however, the positive effects of technological advances are not sufficient to counteract the enormous increase in the use of the car and the growing number of cars in circulation.
In the context of the need to reduce harmful emissions, using large scale coating of titanium dioxide is of particular interest, because its fotocatalítico effect causes the disintegration of the harmful substances in the air.
The various practical applications, such as the covering of facades and streets, as well as the painting of interior spaces, have not succeeded until now fill completely the expectations with regard to the improvement of the quality of the air. Selective modification of titanium dioxide, achieved by means of doping, its effectiveness extends significantly, which considerably improves the intensity of the disintegration of harmful substances.
Disintegration of the harmful substances with the help of the light
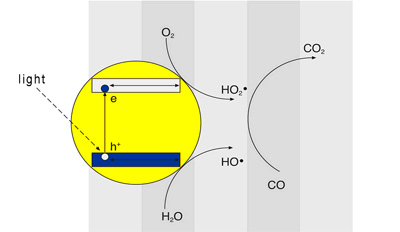
The semiconductor photocatalysis is known since a long time as an efficient method for the transmutation of harmful substances into harmless products, through solar energy. It is based on the capacity of capturing free radicals generated by the absorption of light in the semiconductor surface and use them for electron transfer reactions.
The model of energy bands are used for a detailed description of the basic course of reactions triggered by the action of light. This model describes the energy state of the electrons of the atoms in a crystalline solid. Interactions between the atoms here lead to an expansion of the energy States, i.e., electrons are joined each other in dense way, forming bands of energy: the valence (VB) band and the conduction (CB) band. These energy bands are separated between si by energy fields, which are not electrons (band gap). The size of the band gap determines if, by thermal stimulation or otherwise, the electrons can be transferred from the valence band to the conduction band.
Semiconductors such as titanium dioxide have a band gap so small that the electrons can be transferred through a reduced absorption of energy. The electrons transferred in the valence band to the conduction band form the so-called "pairs electron - hollow". These are the basis for the reactions of disintegration of harmful substances, represented below. With the addition of an external atom (doping), the number of electronhole pairs can significantly increase with which the catalytic effect can be used on a spectrum of waves considerably more.
Greater efficiency through a band banned smaller
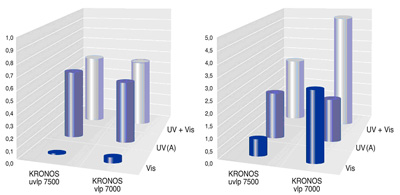
For non-doped titanium dioxide, necessary wavelength for the excitation of the electrons is situated in the UV. This is of 390 nm for the anatasa and 415 for the Roach. Therefore, by titanium dioxide photocatalysis develops especially effectively, when radiation is available exactly at that wavelength. Given that the field UV constitutes only a fraction of the sunlight and artificial light is not available, the use of as a photocatalyst titanium dioxide was limited so far.
Recent research focuses on the possibility of reducing the size of the band gap of the anatasa through the installation of outer atoms in the crystal lattice, and in this way, to take advantage of the catalytic effect on a significantly larger wavelength spectrum. For the doping of titanium dioxide, carbon was appropriate as external atom. Its incorporation reduced the band gap of 3.2 (pure anatasa) eV to 2.32 (phase dopada-carbon), thus, necessary wavelength for the excitement was increased to 535 nm. Thus, titanium taken drugs with carbon dioxide has an effectiveness significantly higher (ultra violet light and visible light > 400 nanometers) than the conventional photocatalysts.
Improvement of the air by the reduction of nitrogen oxides
This statement is confirmed by comparison of the disintegration of the oxides of nitrogen, with taken drugs and not taken drugs titanium dioxide. Through the survey conducted for this purpose, it was investigated how a photocatalyst irradiated with UV (A) or visible light (Vis) disintegrated 1 ppm of nitrogen monoxide in a stream of gas of three litres per minute. The measurement of the concentration of nitric oxide, after the fotocatalítica reaction, was achieved with the help of a system of detector of fluorescence (dissolution 1 ppb). Then, the following results were determined for the disintegration of the nitric oxide (NO). The results of measurement represented below show that the effectiveness of the disintegration of nitric oxide with help of titanium taken drugs with carbon dioxide can be increased considerably, especially through the reinforced effect of the visible part of the spectrum of light. Based on these laboratory results and considering the free conditions, it was determined the potential disintegration of the NO and shown on the right side of the figure.
As the use of as a photocatalyst titanium dioxide is no longer linked to the existence of ideal lighting conditions, numerous harmful substances to the air can fall apart. Materials with coatings photo catalytic active, such as elements of facades, tiles or floor plates lead to the desired reactions of disintegration, including in areas in the shade as the north side of the buildings or tunnels and subwaysas well as under the trees, but also tile, carpet or interior finishes with diffused light.
The application of doped titanium dioxide is an important cornerstone for the fulfilment of the commitment of the industrialized countries reduce emissions of greenhouse gases by 5 per cent by the year 2012, in accordance with the Kyoto Protocol.